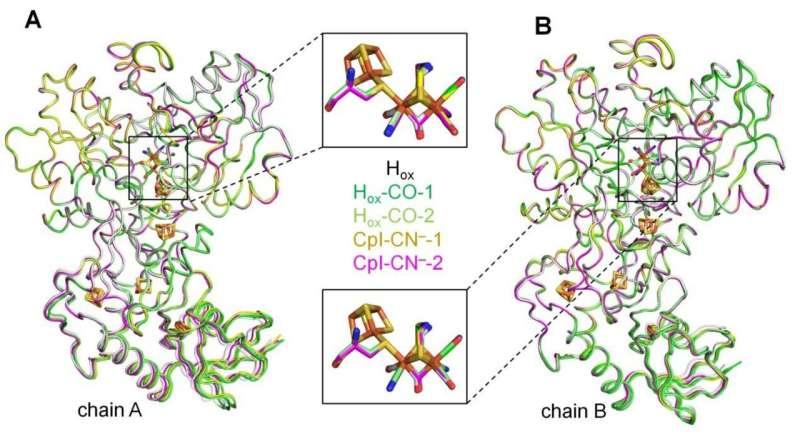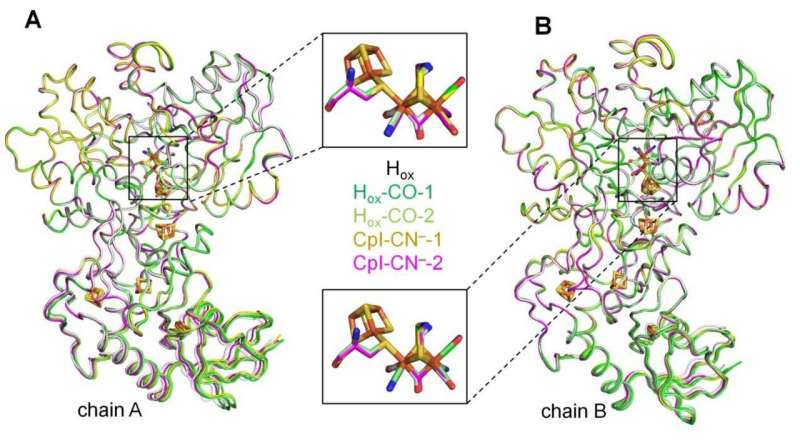
In nature, enzymes termed hydrogenases are capable of producing molecular hydrogen (H2). Special types of these biocatalysts, so-called [FeFe]-hydrogenases, are extremely efficient and therefore of interest for biobased hydrogen production. Although scientists have learned a lot about how these enzymes work, many details remain to be completely understood.
A research team of the Photobiotechnology group at Ruhr University Bochum, Germany, headed by Dr. Jifu Duan and Professor Thomas Happe succeeded in filling a scientific gap. The researchers showed that external cyanide binds to the [FeFe] hydrogenases and inhibits hydrogen formation. In the process, they detected a structural change in the proton transport pathway, which helps to understand the coupling of electron and proton transport. They reported their findings in the journal Angewandte Chemie of December 4, 2022.
A sophisticated internal catalyst
To generate H2, these biocatalysts transfer electrons to protons, employing a sophisticated structure as internal catalyst. This so-called H-cluster contains electronically active iron ions that are bound to what most people know as toxins: carbon monoxide and cyanide.
However, although internal carbon monoxide and cyanide are crucial for the high activity of hydrogenases, additional external carbon monoxide binds to the H-cluster and prevents its H2 production. “Interestingly, cyanide is also a well-known inhibitor of iron-containing biocatalysts,” says Jifu Duan. “And yet, its effect on [FeFe]-hydrogenases has hardly been analyzed before.”
The Bochum-based research team closed this scientific gap. The researchers showed that external cyanide binds to and inhibits [FeFe]-hydrogenases. In collaboration with Professor Eckhard Hofmann, head of the protein crystallography group at RUB, the team obtained the structure of H2-producing biocatalysts to which external cyanide was bound.
“The high-resolution structure in combination with spectroscopic analyses tells us that the external cyanide directly binds to the H-cluster, similar to other inhibitors studied so far,” says Jifu Duan. “This explains why the hydrogenase is inactive after cyanide treatment.”
Coincidental capture of a transient state
When the researchers took a detailed look into the structure of the cyanide-poisoned hydrogenase, they found a surprise. They observed structural changes in the proton transport pathway that is required to guide the protons that will become H2 to the H-cluster.
“This conformation has been suggested to be vital for efficient proton shuttling, but it had never been observed structurally. Coincidently, the cyanide binding helped us to capture such a transient state,” says Jifu Duan.
“These findings are important for researchers to understand the coupling of electron and proton transport which is not only relevant for H2-generating enzymes, but many additional biocatalysts,” concludes Thomas Happe.
More information: Jifu Duan et al, Cyanide Binding to [FeFe]‐Hydrogenase Stabilizes the Alternative Configuration of the Proton Transfer Pathway, Angewandte Chemie International Edition (2022). DOI: 10.1002/anie.202216903
Journal information: Angewandte Chemie International Edition , Angewandte Chemie
Provided by Ruhr-Universitaet-Bochum