The environmentally harmful greenhouse gas carbon dioxide, or CO₂ for short, can be converted into valuable chemical products such as carbon monoxide (CO) or ethanol by means of electrochemical reduction—electrolysis. These can be used as raw materials for industry or for the sustainable provision of energy. However, a key obstacle to the long-term stability of this technology is the water and salt management within the electrolytic cell in which the chemical reaction takes place.
The research team led by Dr. Joey Disch and PD Dr. Severin Vierrath from Hahn-Schickard and the University of Freiburg, in collaboration with the French Institut Laue-Langevin in Grenoble, has made significant progress in understanding the distribution of water during CO₂ electrolysis. Their study was first published in the ACS Energy Letters and has now been featured as a Research Highlight in the February issue of Nature Catalysis.
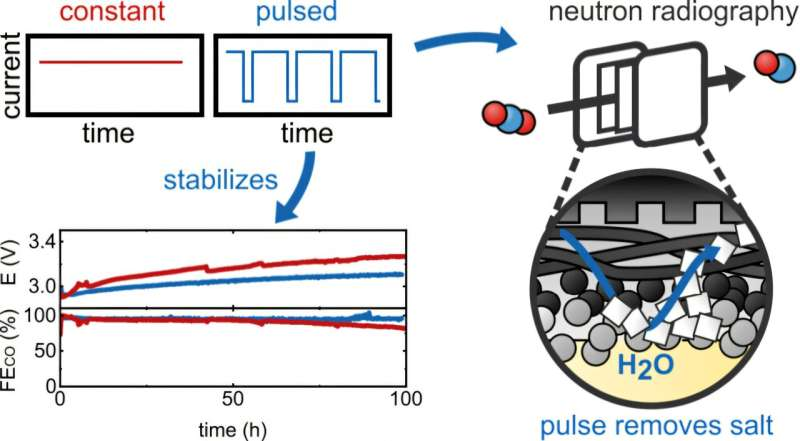
The study uses high-resolution neutron imaging—one of the most powerful methods for directly investigating water transport in electrolyzers—to visualize the transport mechanisms during the pulsed operation of a CO₂ electrolyzer.
With a spatial resolution of 6 μm, this method allows a highly precise investigation of water distribution and salt formation under realistic operating conditions (400 mA cm⁻² at a cell voltage of 3.1 V and a Faradaic efficiency for CO of 95%). In contrast to X-rays, neutrons easily penetrate even metallic components, while making hydrogen and thus water-containing structures highly visible.
The results show a significant stabilization of the electrolyzer during pulsed operation, in which the cell potential is periodically set to a potential below the onset of reduction for a short time. Neutron imaging provides an explanation for the stabilization and illustrates that during the brief interruptions in operation, the water content in the gas diffusion layer increases, which promotes the breakdown of obstructive salt deposits.
Electrochemical CO₂ reduction opens up promising prospects for a sustainable transformation of the chemical industry. In particular, CO₂ electrolysis for the production of carbon monoxide is on the threshold of industrial application: Electrolysis cells with anion exchange membranes are already impressing with remarkable efficiency thanks to optimized reactant management and minimized resistance losses.
These findings thus provide valuable information for optimizing the design and operation of CO₂ electrolyzers, enhancing their efficiency and long-term stability, and facilitating the removal of the harmful greenhouse gas CO₂ from the environment.
More information: Luca Bohn et al, High-Resolution Neutron Imaging of Water Transport in CO2 Electrolysis during Pulsed Operation, ACS Energy Letters (2025). DOI: 10.1021/acsenergylett.4c03003
Marçal Capdevila-Cortada, Pulsed electrolysis through neutron lenses, Nature Catalysis (2025). DOI: 10.1038/s41929-025-01305-w
Journal information: Nature Catalysis , ACS Energy Letters
Provided by Hahn-Schickard
