by Ecole Polytechnique Federale de Lausanne
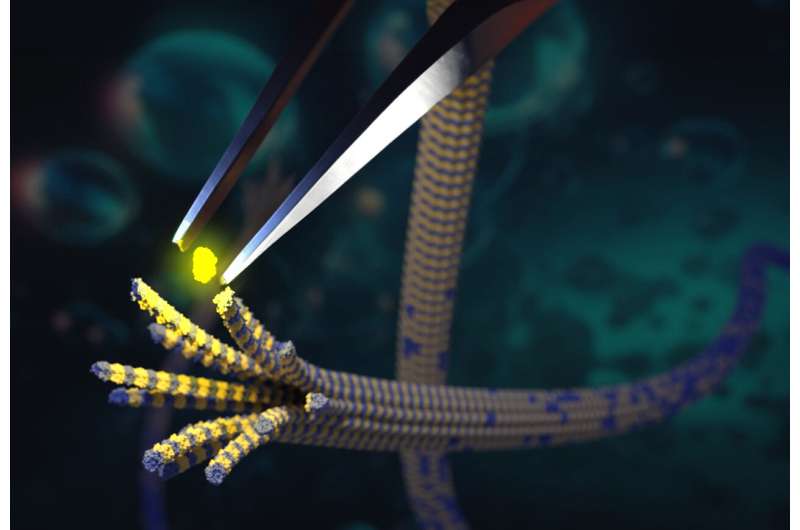
Tubulin is a protein that plays a crucial role in the structure and function of cells. It is the main component of microtubules, which are long, hollow fibers that provide structural support, help the cell divide, give it its shape, and act as tracks for moving molecular cargo around inside the cell.
There are two types of tubulin: alpha-tubulin and beta-tubulin. Together, they form dimeric (two-part) building blocks, spontaneously assembling into microtubules that undergo further continuous cycles of assembly and disassembly.
To fine-tune microtubules, the dimers undergo various post-translational modifications (PTMs), which are chemical modifications that occur after they are synthesized, and can affect their structure, activity, and interactions with other molecules.
Two important PTMs take place on the unstructured tail of alpha-tubulin: Polyglutamylation, which adds chains of glutamate amino acids, and detyrosination, which removes the final tyrosine amino acid. These PTMs, among others, are found together in stable microtubules, e.g. in neurons.
Combinations of PTMs form what scientists refer to as a “tubulin code,” which is connected to specific functions of microtubules. Tubulin PTMs are critical for the proper functioning of microtubules.
Dysregulation of PTMs have been linked to various diseases, including cancer, neurodegeneration, and developmental disorders. Therefore, understanding the importance of tubulin PTMs is crucial for advancing our knowledge of these diseases and developing potential therapies. The problem is that the mechanisms that govern such PTM patterns are not well understood, mostly because we don’t have the tools to dissect the function and regulation of tubulin PTMs.
Cracking the tubulin code
Scientists at EPFL and the University of Geneva (UNIGE) have now developed a chemical method to engineer fully functional tubulin carrying precise combinations of post-translational modifications (PTMs). The study was led by Professors Beat Fierz (EPFL) and Assistant Professor Charlotte Aumeier (UNIGE), in collaboration with the labs of Pierre Gönczy (EPFL) and Carsten Janke (Institute Curie), and provides insight into how specific PTMs regulate the function of tubulin in cells. Their findings have been published in Nature Chemistry.
The method uses chemo-enzymatic protein splicing to attach synthetic alpha-tubulin tails that were modified with varying degrees of polyglutamate to human tubulin molecules. Using these “designer” tubulins allowed the researchers for the first time to assemble homogenously modified microtubules.
The researchers also found that polyglutamylation of alpha-tubulin facilitated its detyrosination by enhancing the activity of the protein complex vasohibin/SVBP, the key enzyme responsible for this modification. The team confirmed their findings by changing the levels of polyglutamate in living cells and observing the effects on tyrosine removal.
The study presents a novel approach to designing tubulins with specific PTMs and uncovers a new interplay between two key regulatory systems that control the function of tubulin: polyglutamylation and detyrosination.
The new method of producing tubulins with defined PTMs can advance our understanding of their molecular function, and provide insights into how dysregulation of these PTMs leads to diseases.
Based on this work, the labs of Fierz and Aumeier, together with Jens Stein at the University of Fribourg and Michael Sixt at ISTA Vienna will next investigate how tubulin PTMs control the cytoskeleton in migrating immune cells.
More information: Eduard Ebberink et al. Tubulin engineering by semi-synthesis reveals that polyglutamylation directs detyrosination, Nature Chemistry (2023). DOI: 10.1038/s41557-023-01228-8. www.nature.com/articles/s41557-023-01228-8
Journal information: Nature Chemistry
Provided by Ecole Polytechnique Federale de Lausanne