In recent years, many engineers and material scientists have been trying to develop sustainable energy solutions that could help to mitigate climate change on Earth. This includes carbon capture technologies, which are specifically designed to capture or absorb carbon dioxide (CO2) in sites where it is widely produced, such as power generation plants or industrial facilities that rely on biomass or burning fossil fuels.
While some carbon capture solutions achieved promising results, those based on conventional wet chemical scrubbing methods utilizing sp3 amines often consume too much energy and are prone to corrosion and sorbent degradation. This significantly limits their widespread implementation, highlighting the need for alternative CO2 separation strategies.
Researchers at Johns Hopkins University, the University of Texas at Austin and Massachusetts Institute of Technology (MIT) have recently introduced a series of redox-tunable Lewis bases (i.e., molecules with a lone pair of electrons that can be donated to a coordinate covalent bond) with sp2 nitrogen centers that can reversibly capture and release CO2. In their paper, published in Nature Energy, they also outline strategies to fine-tune the properties of these Lewis bases.
“We demonstrate a library of redox-tunable Lewis bases with sp2-nitrogen centers that can reversibly capture and release carbon dioxide through an electrochemical cycle,” Xing Li, Xunhua Zhao, Yuanyue Liu, T. Alan Hatton and Yayuan Liu wrote in their paper. “The mechanism of the carbon capture process is elucidated via a combined experimental and computational approach.”
The researchers’ recent work is based on the idea that due to their CO2 affinity, Lewis bases with redox-active sp2 nitrogen centers could be modulated using electrochemical potentials, enabling the development of alternative, more effective carbon capture solutions. To verify this hypothesis, the researchers compiled a library of organic bases containing sp2 nitrogen centers, including pyradine, diazine, thiadizole and azo moieties.
“While in the oxidized form, none of these compounds shows strong interactions with CO2,” the researchers wrote in their paper. “However, their electro-reduction and subsequent oxidation behavior can be drastically modulated by the presence of CO2 in the electrolyte.”
Li and his colleagues clearly outlined the mechanism that allows the Lewis base sorbents to capture carbon in both computational simulations and experiments. Subsequently, they showed that the properties of these sorbents can be fine-tuned (i.e., tailored for specific uses) using both molecular design and electrolyte engineering methods. By fine-tuning the properties of the Lewis bases they created, the researchers were then able to identify a particularly promising Lewis base based on bifunctional azopyridine.
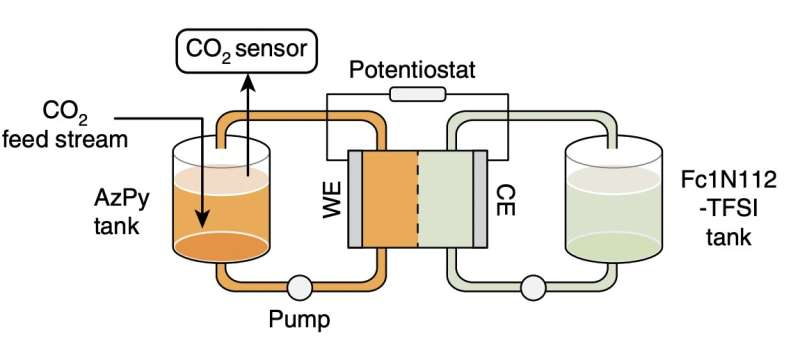
“We identify a bifunctional azopyridine base that holds promise for electrochemically mediated carbon capture, exhibiting >85% capacity utilization efficiency over cycling in a flow system under 15% carbon dioxide with 5% oxygen,” Li and his colleagues wrote in their paper. “This work broadens the structural scope of redox-active carbon dioxide sorbents and provides design guidelines on molecules with tunable basicity under electrochemical conditions.”
In the future, the bifunctional azopyridine base identified by Li and his colleagues could be used to create more energy-efficient and effective carbon capture technologies. In addition, their work could pave the way towards the development of other carbon capture solutions based on Lewis base sorbents.
More information: Xing Li et al, Redox-tunable Lewis bases for electrochemical carbon dioxide capture, Nature Energy (2022). DOI: 10.1038/s41560-022-01137-z
Journal information: Nature Energy
© 2022 Science X Network